Atom, Electron and Electric Charge — Why Electric Current Is Possible

In the previous article, we explained that electricity is the ordered movement of electric charges.
But this leads to a deeper question:
Why can these charges move at all?
Why does current flow easily in a metal wire, but not in wood or plastic?
Why don’t electrons remain permanently “attached” to their atoms?
And what exactly must happen inside matter for a directed flow to appear?
To understand this, we need to look at the structure of the atom.
⚛ The Atom — The Foundation of Matter
Everything around us — air, water, metal, plastic — is made of atoms.
An atom is the smallest unit of a chemical element that still retains its properties.
Every atom consists of:
- a nucleus
- electrons
The nucleus is located at the center of the atom and contains:
- protons — particles with a positive charge
- neutrons — particles with no charge
The number of protons determines the element.
For example, copper differs from iron because of the number of protons in its nucleus.
Surrounding the nucleus are electrons — particles with a negative charge.
In its normal state, an atom is electrically neutral: the number of positive and negative charges is equal.
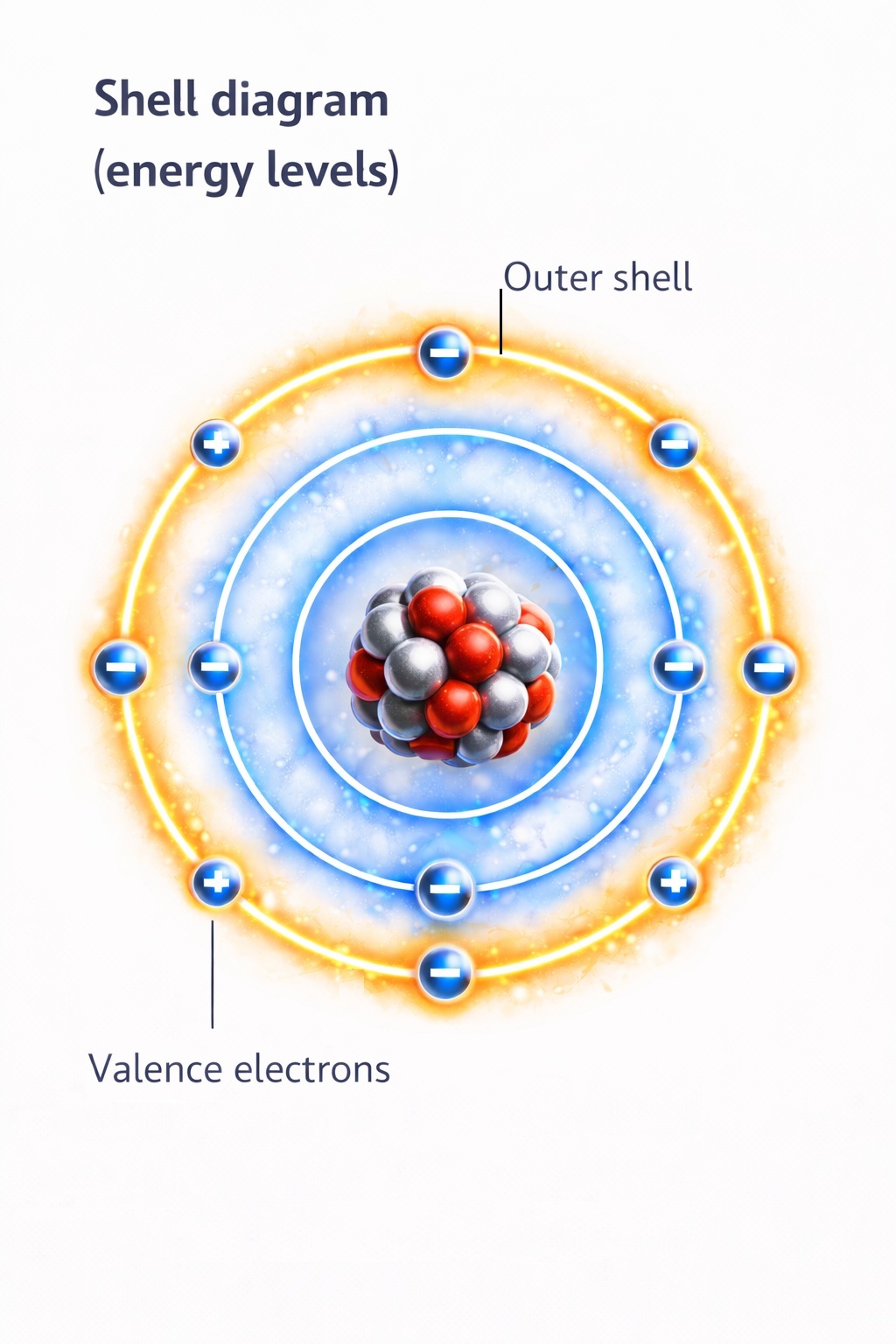
However, what truly matters for electricity is not just the presence of charges, but how strongly electrons are bound to the nucleus.
Why Electrons Are Bound with Different Strength
Electrons occupy specific energy levels (also called shells) around the nucleus.
The closer an electron is to the nucleus, the stronger the attraction.
The farther away it is, the weaker the bond.
The most important electrons for electrical phenomena are those located in the outermost shell. These are called valence electrons.
Valence electrons:
- participate in chemical reactions,
- can move between atoms,
- determine whether a material conducts electricity.
If valence electrons are tightly bound, the material behaves as an insulator.
If they are loosely bound, they can move more freely.
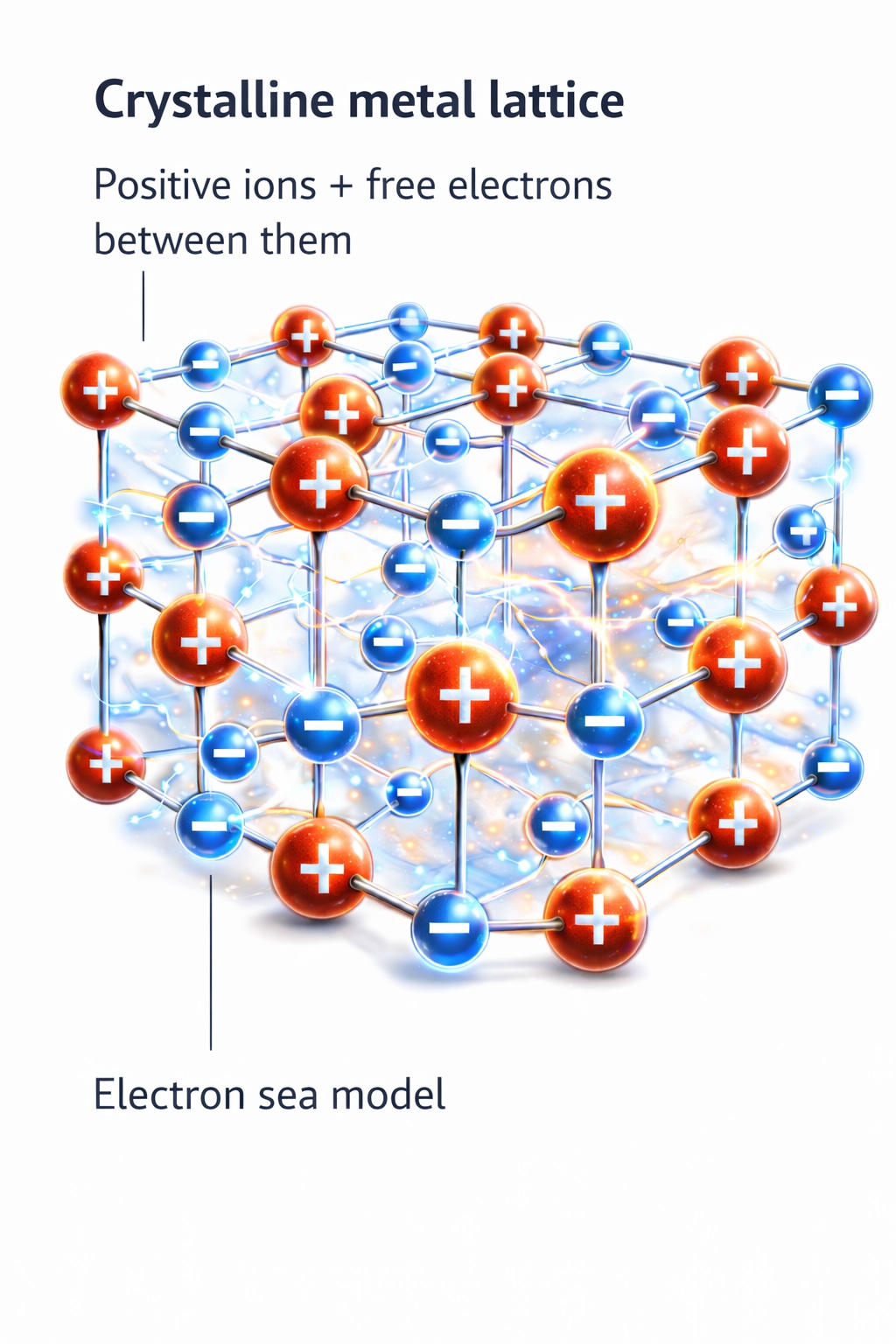
Why Metals Conduct Electricity
In metals, valence electrons are relatively weakly bound.
Metal atoms form an ordered structure known as a crystal lattice.
However, some electrons are not tied to a single atom — they can move freely between them.
You can imagine a framework of positively charged ions with a “sea” of free electrons moving between them.
This means that metals already contain free charge carriers.
When a battery or generator is connected to a metal conductor:
- an electric field is created inside the conductor,
- this field acts on the free electrons,
- electrons begin to drift in one general direction,
- electric current appears.
It is important to understand: electrons do not suddenly appear.
They are already present — the electric field simply organizes their motion.

The Electric Field — What Causes Motion
When a power source is connected, it creates an electric field.
An electric field is a region where electric charges experience a force.
This force causes electrons to move in a directed way.
Without an electric field, electron motion remains random.
With the field, it becomes organized.
This can be compared to wind.
Air molecules are always present, but wind gives them a common direction.
Why Not All Materials Conduct Electricity
In insulators — such as wood, glass, or plastic — electrons are much more tightly bound to their atoms.
There are almost no free electrons.
Even if an electric field is applied, charges cannot move freely.
That is why current does not flow, or flows only extremely weakly, in these materials.
This is also why insulators are used to protect us from electric shock.
Semiconductors — A Special Case
Some materials lie between conductors and insulators. These are called semiconductors.
Under normal conditions, they conduct poorly.
However, when temperature changes, light is applied, or special impurities are added, their conductivity can increase dramatically.
In semiconductors, charge carriers include not only electrons, but also so-called “holes” — positions where an electron is missing.
Controlling the movement of electrons and holes is the basis of:
- diodes
- transistors
- integrated circuits
- processors
Modern electronics is built on this principle.
![]()
Electrons Move Slowly — But Current Acts Quickly
A common misconception is that electrons race through wires at extremely high speeds.
In reality, their drift velocity is very slow — often only millimeters per second.
However, the electric field spreads through the circuit almost instantly.
That is why a lamp lights up immediately after you flip a switch.
It is important to distinguish between:
- the speed of the electric field
- the speed of the electrons themselves
These are not the same thing.
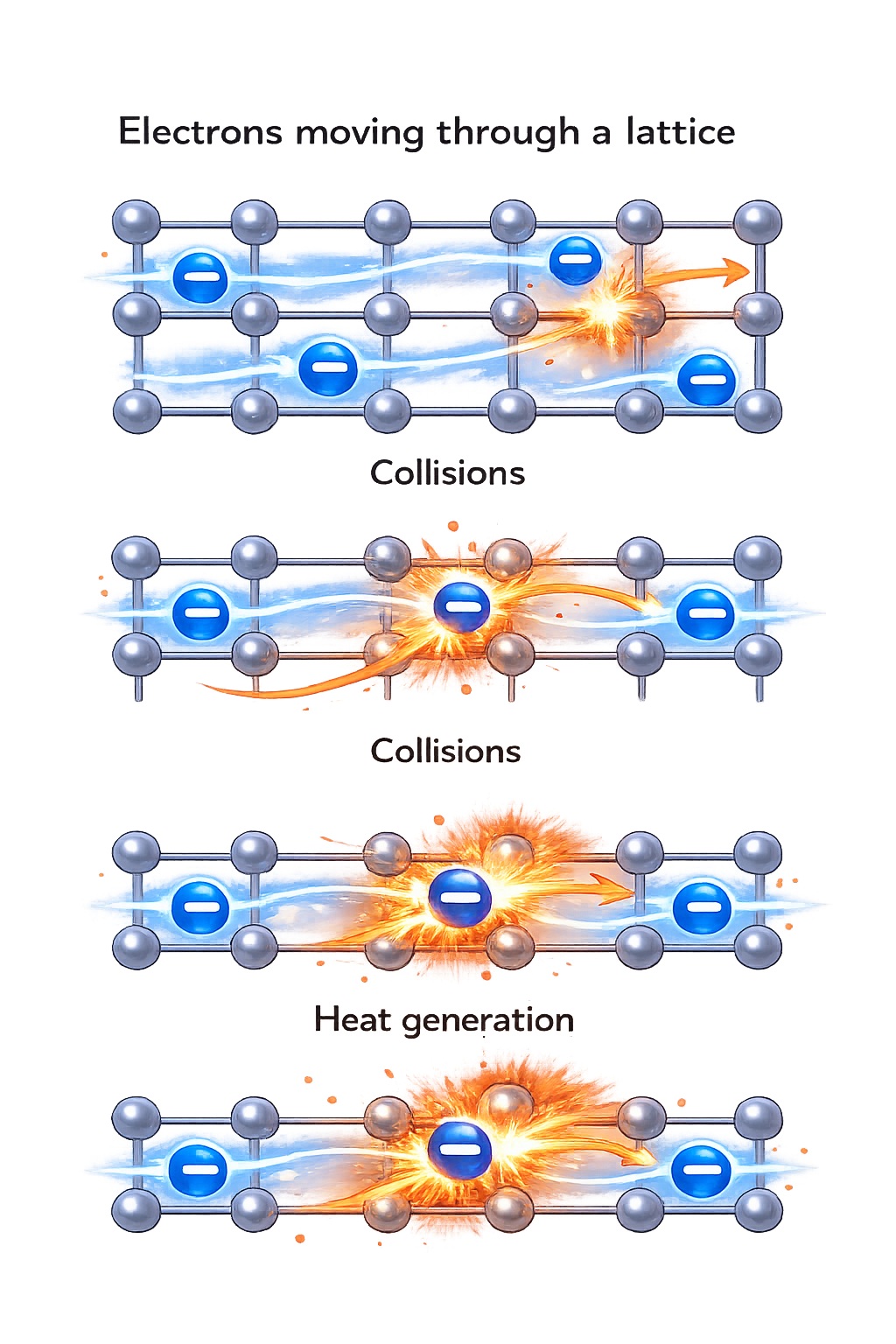
Why a Wire Heats Up
As electrons move through a conductor, they collide with atoms in the crystal lattice.
These collisions produce heat.
That is why:
- a thin wire can overheat,
- high-power devices require thick cables,
- there are limits to how much current a conductor can safely carry.
Heating is a result of resistance opposing the motion of electrons.
The Fundamental Reason Current Is Possible
Now we can bring everything together.
Electric current is possible because:
- Matter is made of atoms.
- Atoms contain electrically charged particles.
- Some materials contain free electrons.
- An electric field can organize their motion.
If any of these conditions were missing, current would not exist.
Electricity is not something added to matter from outside.
It is a natural consequence of how matter is structured.
Conclusion
In this article, we explored:
- the structure of the atom,
- the role of the nucleus and electrons,
- the importance of valence electrons,
- why metals conduct electricity,
- why insulators do not,
- and how the electric field enables current flow.
Electric current is not magic.
It is a natural result of the structure of matter.
Next Article
In the next article, we will answer another fundamental question:
What makes electrons start moving?
We will introduce the concept of voltage and understand why current cannot exist without it.
You can also:
🔧 visit the “Devices” section to see practical applications;
📘 continue learning in the “Education” section;
💬 ask questions or join the discussion on the Forum.